The software is becoming more and more important part of medical devices in the future. That’s why it is very crucial for the companies working in the field to be able to develop even more complex...
Recent Posts
Across industries, the pace to introduce new products is increasing. This puts pressure to shorten the design and development cycles. Medical devices are no exception in this respect.
There’s few common trends in the medical field, which have a clear effect to the development of new medical products. The first one is the constantly changing jungle of rules and regulations, which...
Across industries, the pace to introduce new products is increasing. This puts pressure to shortening the design and development cycles. Medical devices are no exception in this respect.
An agile co-creation of next generation medical devices – Innokas launches the MedTech Design Studio
Innokas Medical renewed its strategy during the last year, aiming to achieve the company’s new vision and to take the changes of the operative environment as well as the development needs of the...
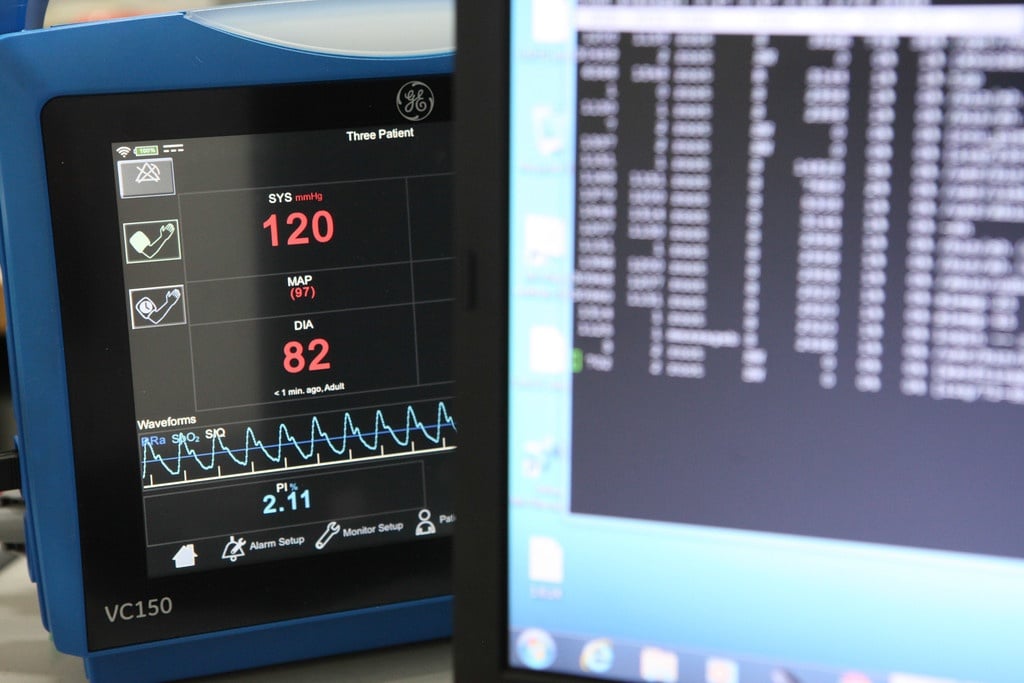
The Early Warning Score (EWS) is a medical solution taking risk management in healthcare to the next level. Innokas Medical and GE Healthcare have now developed the new version of the software used...
Innokas Medical has further reinforced its know-how in medical device design and development during this autumn. Three new resources have joined Innokas’ mechanical and electronics engineering team,...
“At Innokas Medical, everything begins with the quality built into our processes, products and services. The strong know-how we have in-house as well as the high standards of our daily operations can...
Are you ready to roll up your sleeves and show us what you got? Are you willing to challenge yourself and achieve better and better results with high quality medical device products? If you answered...