Innokas Medical will participate in Medico Bazar 2020 -event which will be held in Lyngby, Denmark (in Technical University Library) on 12th March. Medico Bazar displays the newest devices, projects...
Recent Posts
When creating a new medical device to the market, there’s much more that goes into designing, developing and introducing it to the market. To be able to enter the certain markets, the development and...
Innokas Medical will participate in European Healthtech Investment Forum –eventwhich will be arranged in Helsinki, Finlandon 10th to 11th March 2020. The event brings together leading investors...
Information Driven Care in Response to Healthcare Challenges were discussed in Digital Health Nordic
Innokas Medical participated in Digital Health Nordic 2020 -event together with its sister company Cubist IT AB from Sweden. Innokas and Cubist were the main sponsors of the event. Digital Health...
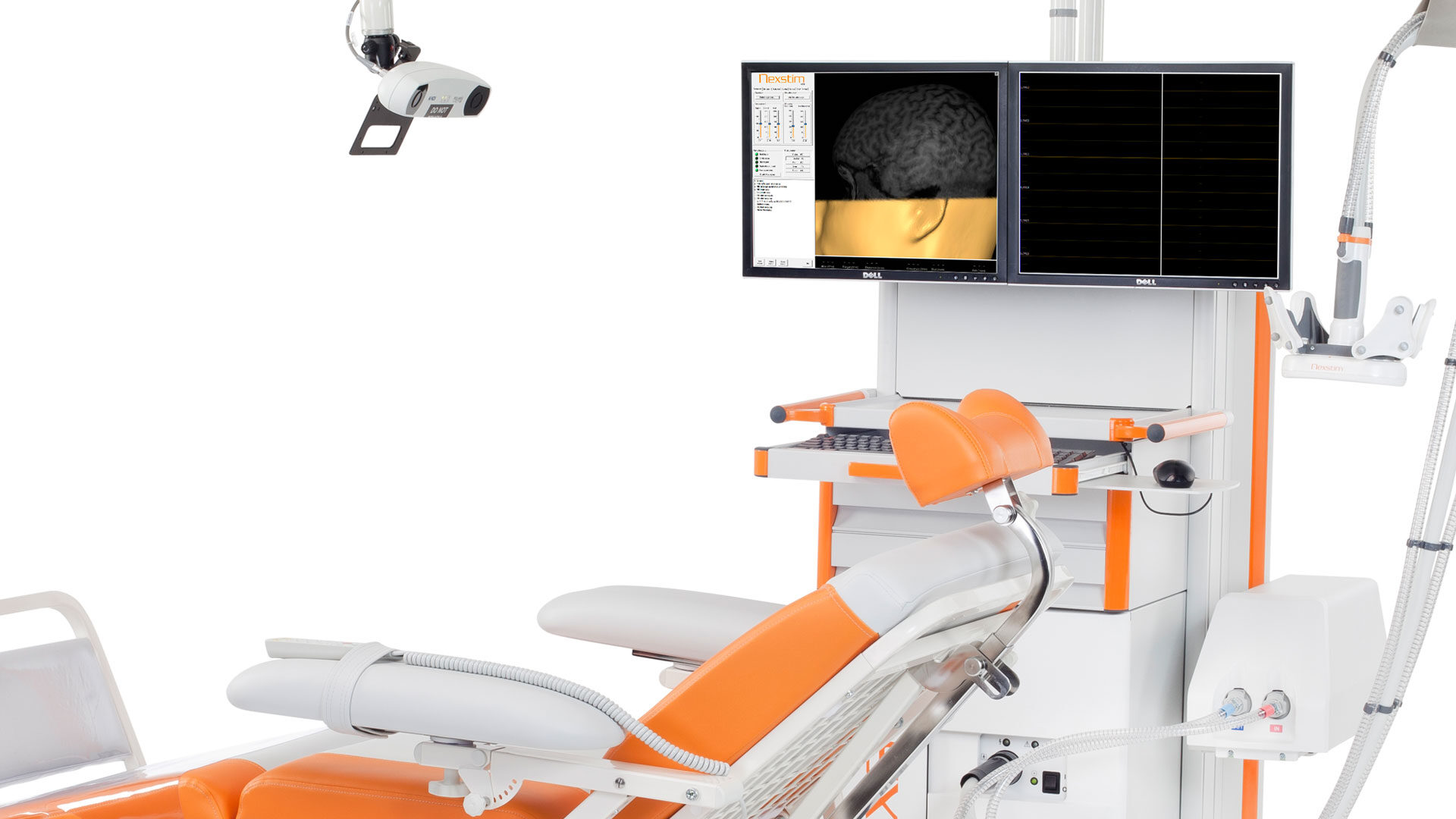
Nexstim Plc. develops and manufactures NBS equipment for demanding brain diagnostics. The NBS (Navigated Brain Stimulation) System combines 3D brain mapping, stimulation coil positioning, and...