Innokas Medical renewed its strategy during the last year, aiming to achieve the company’s new vision and to take the changes of the operative environment as well as the development needs of the organization into account. As part of this strategic development work, Innokas wanted to renew and develop its engineering function as well. As a result of this development work, Innokas has now launched a new business concept, Innokas MedTech Design Studio, which highlights the value add of strategic partnerships: agile development of next generation medical devices will be achieved by co-creation and standardized working methods.
“Our new vision is to be the preferred medtech long-term co-creation partner in 2020. To achieve this, we started to develop our way of working and processes last year”, tells Jenni Tuulos, Head of Innokas MedTech Design Studio.
“One major part of this development work is Innokas’ MedTech Design Studio, which we’ve now launched. With this new business concept we’re able to take the changes and challenges of more complex operative environment into account efficiently, when at the same time ensuring the agile development cycle for next generation medical devices”, she continues.
By more complex operative environment Tuulos means both the constantly changing jungle of rules and regulations but also one common trend that all companies should prepare to: everything is getting much more complicated.
“The complexity is caused by digitalization; different technologies are developed even more rapidly, the impact of software is growing in medical devices as well, and wireless as well as mobility features are increasing. In addition, the user experience and safety issues are even more crucial nowadays”, she argues and continues:
“This all creates a whole new kind of challenges for medical device developers, as companies need to know and, in particular, need to master a significant number of different kind of technologies and platforms already on a very early phase of the product lifecycle. The complexity and the complicated regulations governing healthcare technology cause that designing and developing new medical devices become slower and more expensive. We’d say that this is a very complex equation to solve.”
In practice, we’re speaking of an equation that cannot be overlooked: the changes force also health technology companies to change their mindsets and ways of working in order to run along with the global competition.
Renewed product development model brings agility
Innokas’ answer for this is the MedTech Design Studio concept the company has now launched. Innokas MedTech Design Studio is based on the co-creation mindset highlighting value add strategic partnerships for co-creating next generation medical devices. In co-creation, all parties have a competitive edge and are able to add more value compared to the previous business models, as it brings together all the participants from end-users to experts to develop new products.
In addition to co-creation, Innokas MedTech Design Studio invests in agility. According to Innokas, an agile product development really is possible in medical business despite the burdensome regulations and more complex business environment.
“We started to think how the development model of medical devices should look like in order to take into account both regulations, laws and standards as well as the agile development cycle of new products. The need for this has arisen through our own experiences as well as through conversations with our customers – there is a pressure to get high-quality medical devices quickly to the market, and this pressure is common for everyone in the field. Co-creation mindset is only one answer – in addition, we wanted to apply commonly known methodologies of product development – design thinking, lean startup and agile – to medical environment”, Tuulos tells.
As a result of this brainwork Innokas created new kind of product development model, which is the basic framework for Innokas’ MedTech Design Studio operation. It shows that medical device development can greatly benefit from the three methodologies. However, there are some notable differences that must be accounted for so that these approaches will work seamlessly together and add business value.
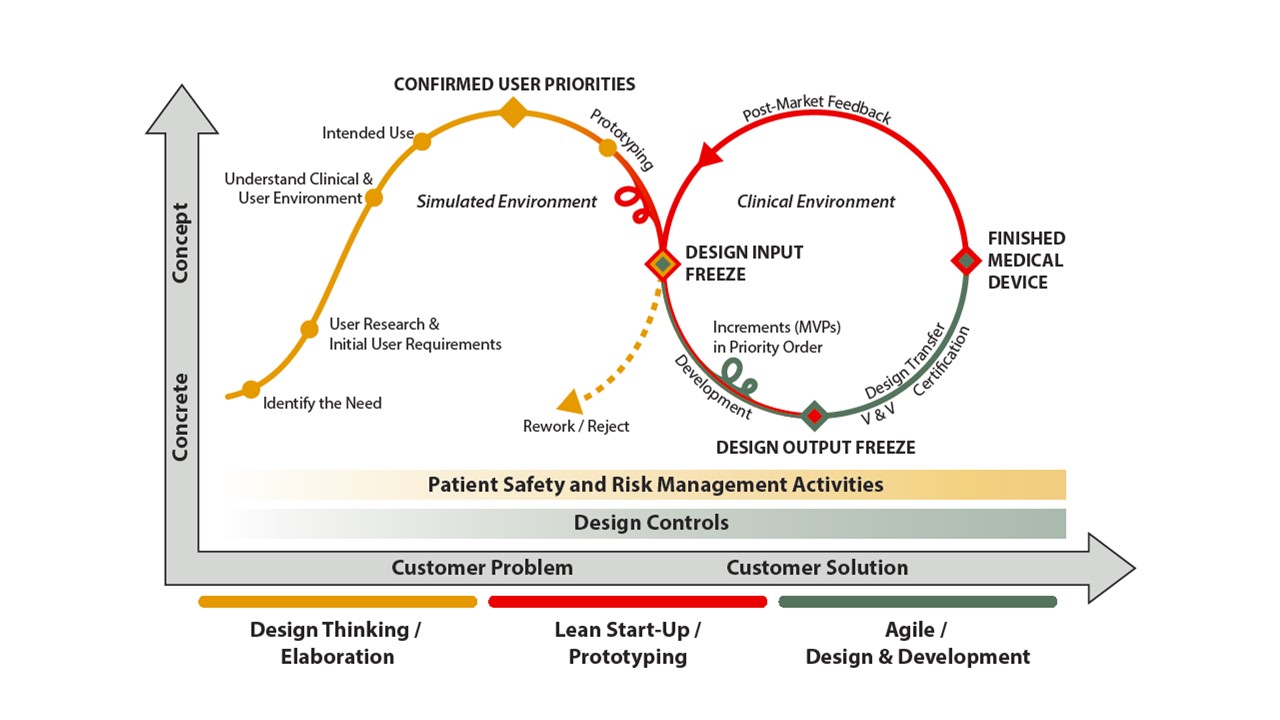
Design Thinking, Lean Start-Up and Agile methods in Medical Device Design and Development. To read more about Innokas MedTech Design Studio’s approach in applying these methods in Medical Device Design and Development, please download your Free White Paper, which can be found in the end of this post!
“When creating this model, we needed to take into account the need for quality management in the field, as well as patient safety and risk management. These determine the design and development of medical devices right from the idea phase, with increasing intensity”, tells Pauli Innamaa, Head of Concepts and Methods at Innokas, who was heading the development of the model at Innokas.
Innokas wants to especially emphasize the idea phase, as with the right choices made already in that phase the more agile project schedules can be achieved.
“The regulations of the whole lifecycle of the product need to be taken into account already in the very early idea phase of the design, because, e.g., the intended use and the classification of the device have a significant impact on the laws, standards and regulations as well as on the needed documentation the authorities require. The devices are more difficult and expensive to be redesigned if the product is already close to launch, which naturally has an impact on the budget that might be exceeded and launching schedule will be overdue”, Innamaa states.
Prototyping is also emphasized in this new, more agile product development model: when the user experience, technological choices as well as the manufacturability of the product are studied already at the product development phase, it will lead to more agile project schedules as well.
”Both prototypes and minimum viable products are used to ensure those aspects already at the early product development phase”, Innamaa says.
Wide competence pool as a competitive edge
In addition to the new product development model, Innokas wanted to refresh the service offering of Innokas MedTech Design Studio. The new service offering is based on certain packages designed and targeted for each product development phase. At the idea phase, Innokas offers consultation with concept clarification and regulatory and risk impact assessment; in the prototyping phase the proof of the concept is achieved, including first prototypes and input crafting; and the design and development phase includes actual design implementation and design transfer.
“We see that we have more clear and flexible service offering now, and it can be better tailored to meet our customers’ specific requirements. We can work as our customers’ strategic partner in some specific phases of the development cycle, or even through the whole product development process of the product, from idea conceptualization through device development and project implementation to transfer to production”, Tuulos says.
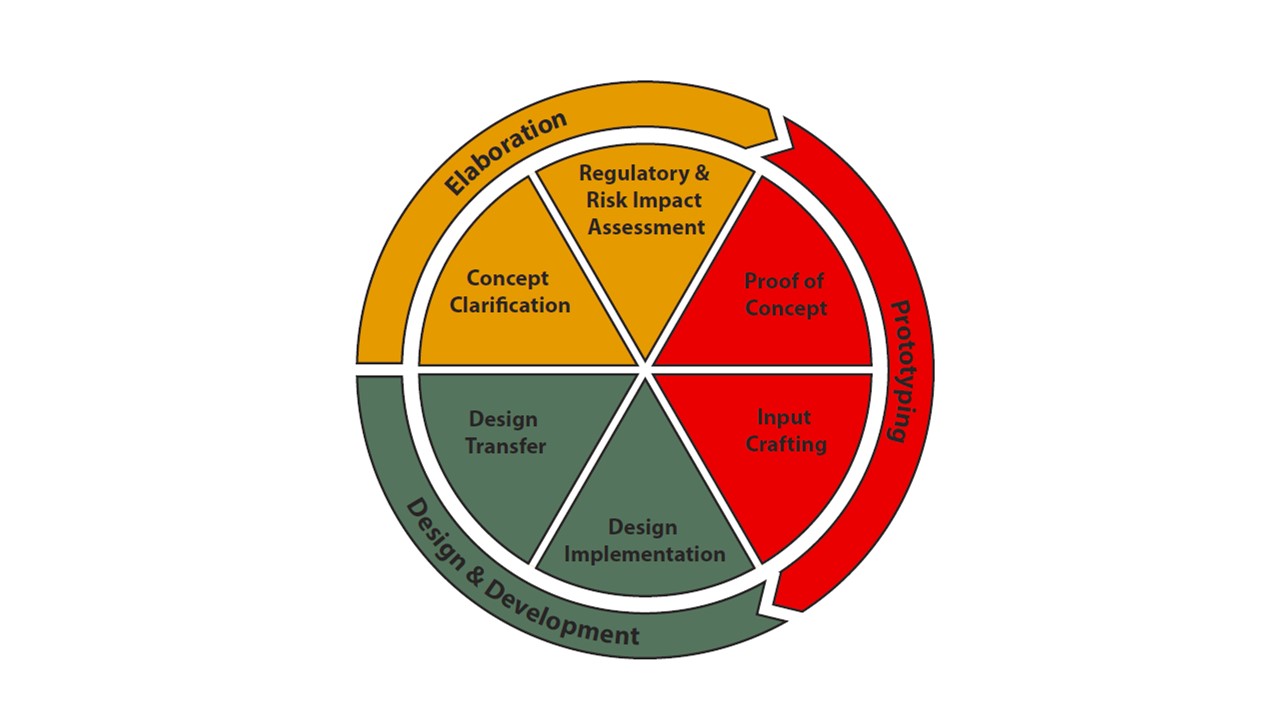
Innokas Medical has created certain service packages which are designed and targeted for each product development phase.
Tuulos believes that the new kind of business concept Innokas has now launched brings added value for Innokas’ customers.
“Our highly skilled MedTech Design Studio team includes +60 employees from all the necessary competence areas of product development, such as electrical and mechanical engineering, embedded software development, industrial and usability design, tester development, prototyping, verification and validation as well as quality, risk and project management. By choosing to develop a new innovation with Innokas MedTech Design Studio team, our customer gets access to a wide spectrum of competences as well as to new kind of agile product development environment”, Tuulos says and emphasizes:
”Our uniqueness, however, lies in our comprehensive service offering, which, in addition to our MedTech Design Studio offering, includes versatile manufacturing and life cycle management services, as well as our strong experience in quality and regulatory affairs. The concept is superior for companies that need long-term co-creation partnerships for the product's whole lifecycle.”











