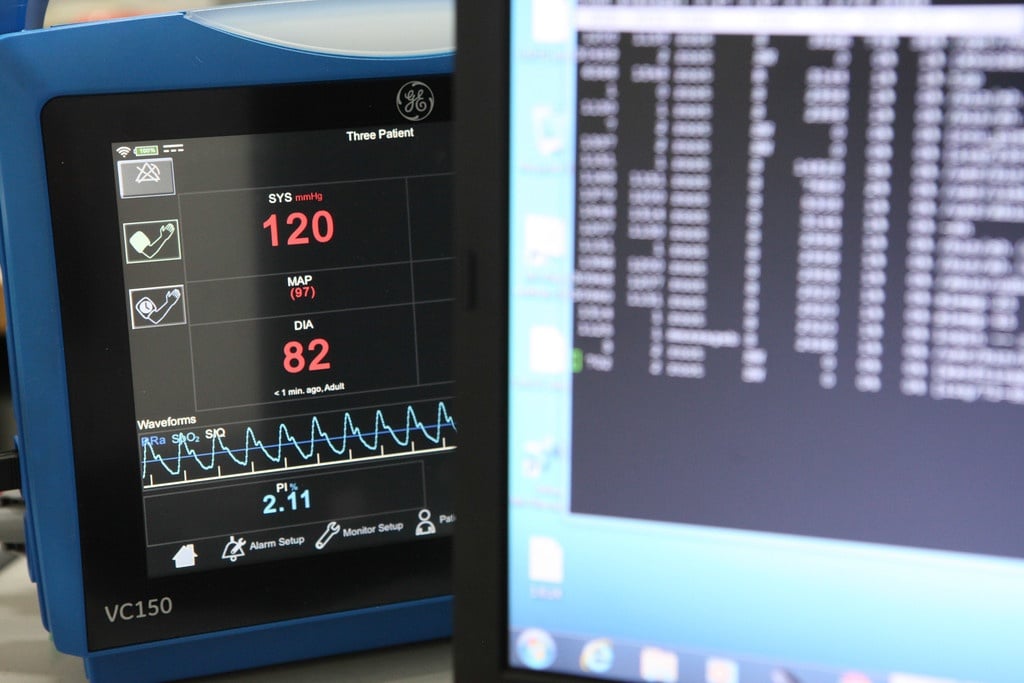
Healthcare technology is one of the largest high-tech export segments of Finnish industry, and it continues to grow strongly year after year. In addition to growth, the industry is evolving with...
Recent Posts
The national Dream Workday is coming again on 2nd October with aim to bring positive energy to the workday and to promote well-being at work. The theme of the day at Innokas is training and recovery...
Innokas Medical has launched free webinar series to share ideas and experiences around medical device quality and regulatory matters. In our sixth webinar we will discuss about MDSAP program and...
Innokas Medical and its personnel participated in the Cycling Competition (Kilometrikisa in Finnish), which was a playful contest aiming to enthuse people to cycle more. The Cycling Competition...
Through MDSAP (Medical Device Single Audit) certificate, the companies are able to indicate they operate according to ISO 13485:2016 -standard and regulatory requirements set by Australia, Brazil,...
Innokas Medical and Serres are sponsoring one of the 14 guest rooms of Ronald McDonald House (RMH) Charities in Meilahti, Helsinki. Sponsorship helps covering the costs of operating the House,...
Innokas Medical and our sister company in Sweden, Cubist IT AB, will participate in the Health100 -event, which will be organized by Upgraded, a non-profit association for health & wellbeing startups...
Innokas Medical launches free webinar in series around the topic “Postponed MDR – how to get the best out of upcoming 12 months.” In our fifth webinar we will discuss about ISO 13485 compliance....
Innokas Medical will participate in the Helseteknologikonferansen -event, which will be organized by Norway Health Tech, Tekna and Norwegian Smart Care Cluster on September 10th. The event will be...