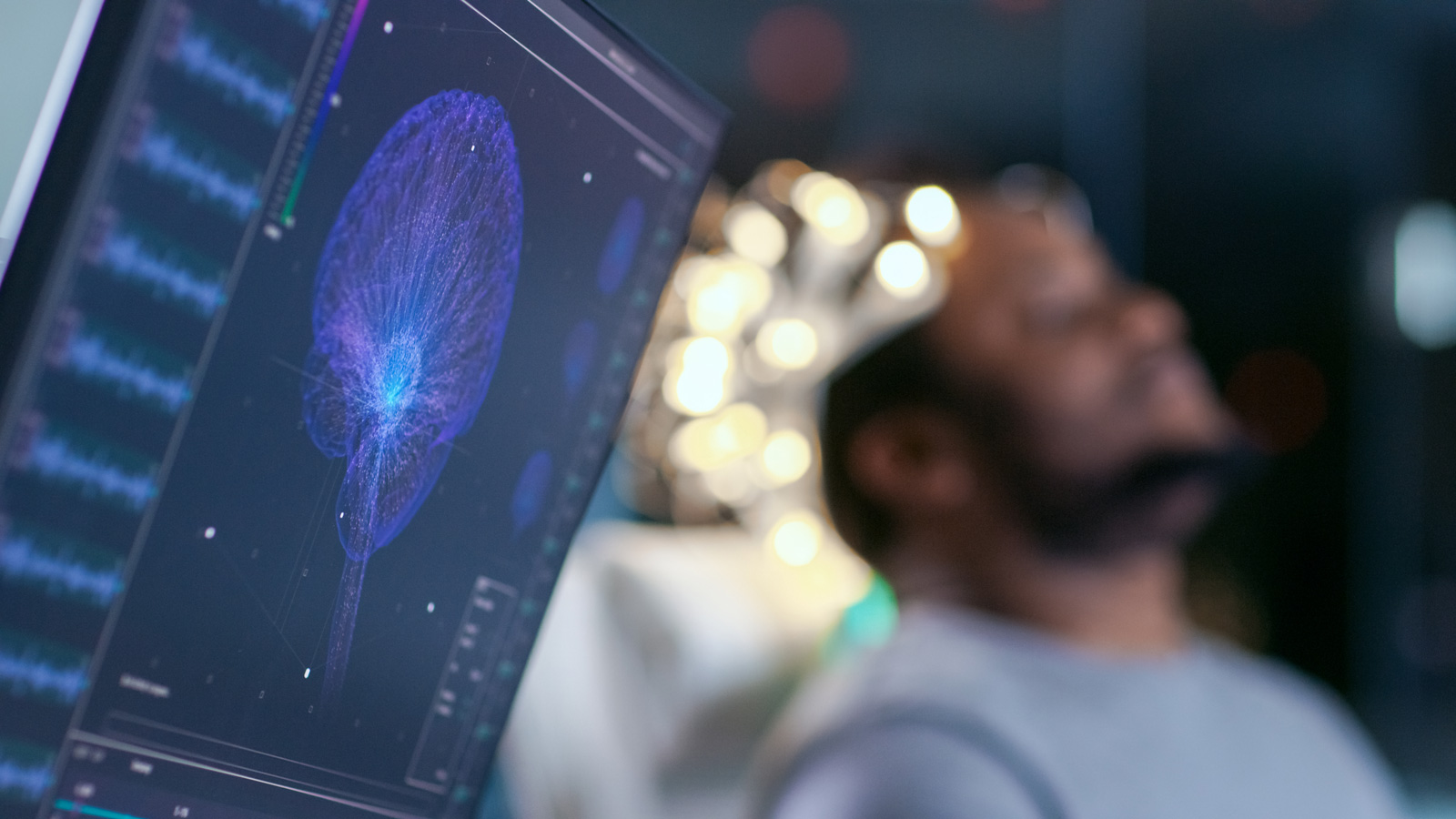
Efficient cooperation between expert partners and well-fitted practices and tools can significantly impact the time to market, reduce costs and simplify the product development of complex solutions.
Recent Posts
It’s a remarkable day for Innokas; our first ever sustainability report has been made public!
To succeed in taking a medical device product from idea to the market, both the design and development flow, as well as the regulatory flow, need to be considered. As the inventor or manufacturer of...
In the manufacturing industry, good procurement can have a major impact on product costs, ease of designing and manufacturability. But how’s the case now as the world situation changed unpredictably...
Innokas Medical, a Finnish technology company specializing in medical technology, is launching an outsourced responsible manufacturer service for companies striving to reach the highly regulated...
Terveystalo is the first Finnish healthcare service company to be granted the ISO 13485 quality management system certificate. The company invests heavily in the product development of intelligent...
How do you solve the key challenges in turning a demanding technical solution idea into a tangible product and bringing it to the market? Could a capable contract designer and manufacturer help you...
Innokas Medical has joined the Finnish HL7 organisation in February 2022. Thus Innokas and especially its software house Digious continue to deepen their cooperation with the key industry players...
The 2022 Health Valley event is starting on March 15th and is accessible both live and online. The three-day healthcare innovation conference is taking place in Nijmegen, the Netherlands, collecting...