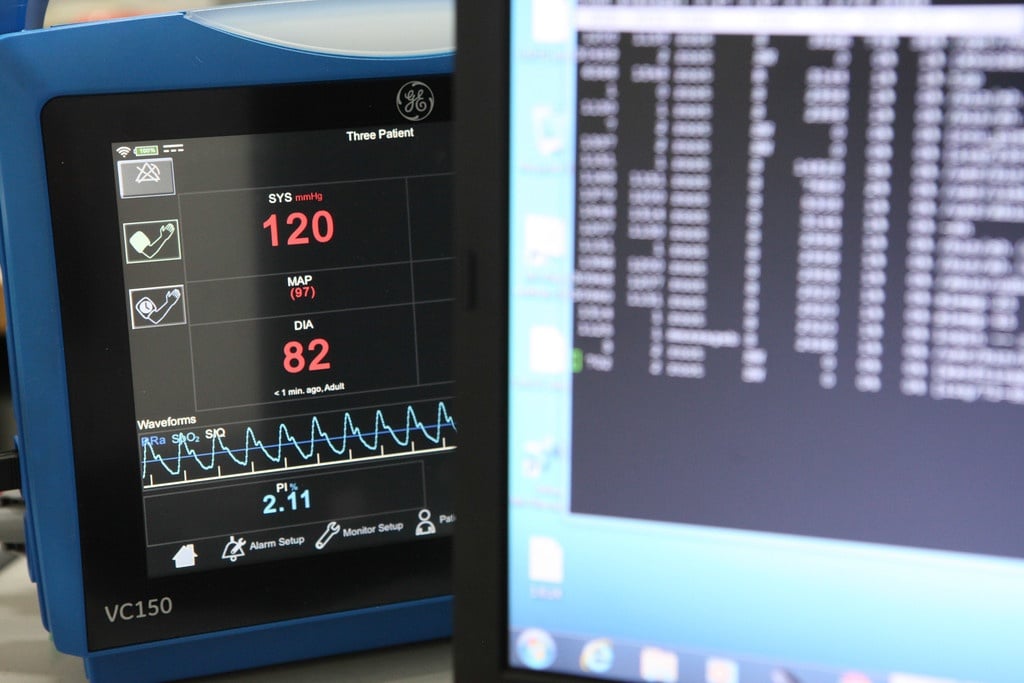
The hot topics in HIMSS (Healthcare Information and Management Systems Society) Europe exhibition in Helsinki were human centricity and how to enable easier connectivity and interoperability with...
Recent Posts
Last year Innokas launched a new strategy to seek strong growth. This means we are extending our presence into new market areas as well as developing our services and launching new ones. In 2022 we...
Innokas Medical, a Finnish technology company specializing in medical technology, is launching an outsourced responsible manufacturer service for companies striving to reach the highly regulated...
Last summer, Lybe Scientific got its first product registered as a CE-marked IVD medical device fulfilling the requirements set by In Vitro Diagnostic Directive (IVDD). The company built an ISO 13485...
DC-Air™ is an intraoral x-ray imaging sensor designed, developed, and manufactured by a Finnish company Athlos. In July 2021, the sensor passed the FDA 510(k) clearance and is now legally permitted...
Long-awaited moment finally happened in May, as after a one-year delay the European Union's new Medical Device Regulation (MDR) came into force. We at Innokas Medical are well-prepared for the MDR...
Innokas Medical participated in the project where the regulatory framework for NTNU’s new set of reagents were created.
Medical device regulations include requirements with which both the device and their manufacturer need to comply to enable legal market entry. However, these requirements are often vaque, described...
Healthcare technology is one of the largest high-tech export segments of Finnish industry, and it continues to grow strongly year after year. In addition to growth, the industry is evolving with...