The collaborative consortium has received a significant funding from Business Finland. This two-year Stroke-DATA -project, which is one part of Business Finland’s Smart Life program, is carried out...
Recent Posts
Norwegian healthtech innovator Augere Medical AS has selected Innokas Medical as their co-creation partner to offer quality and regulatory as well as design and development support to bring their...
When creating a new medical device to the market, there’s much more that goes into designing, developing and introducing it to the market. To be able to enter the certain markets, the development and...
The importance of SW is growing – also in medical devices and as medical devices. This blog text deals with the change of SW’s role, the tightening regulations landscape as well as how to be agile...
Danish company Samplix ApS selected Innokas Medical to co-create the novel Xdrop™ instrument for droplet-based PCR-free enrichment of genomic regions.
“At Innokas Medical, everything begins with the quality built into our processes, products and services. The most important know-how as well as the high standards of our daily operations can be seen...
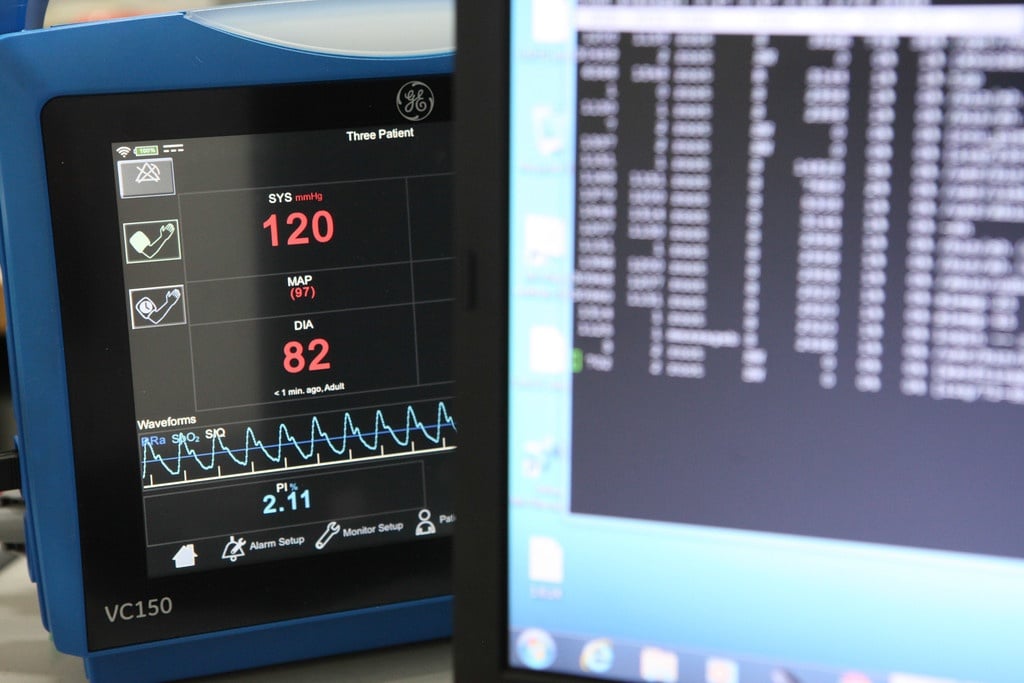
Medical devices have remarkably important role in patient care. The key element in the sector is that there cannot be any compromises done with the safety and effectiveness of the devices – the...
Innokas Design Studio develops software for regulated medical devices to improve peoples' well-being
Healthcare technology is one of the largest high-tech export segments of Finnish industry, and the industry continues to grow strongly year after year. In addition to growth, development is also...