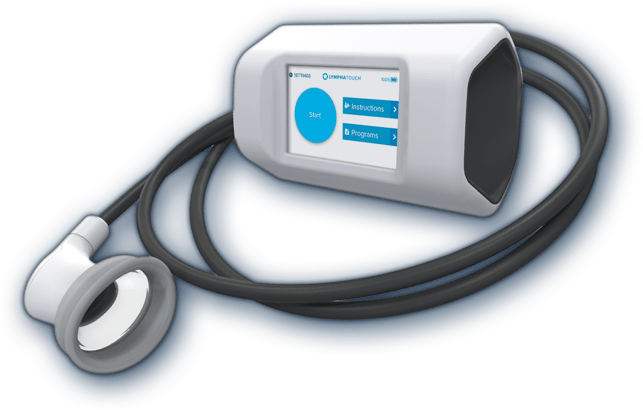
The objective of Finnish healthcare technology company Lymphatouch’s negative pressure treatment is to improve patients’ quality of life by reducing pain and tissue swelling. Innokas Medical is involved in this mission by manufacturing LymphaTouch® devices in the company’s Kempele factory.
Lymphatouch Oy (formerly known as HLD Healthy Life Devices Oy) is a Finnish healthcare technology company, which develops medical devices for faster and easier recoveries. The company’s devices are being used both in hospitals and medical clinics globally.
“Our devices are used daily by, for example, physiotherapists, caregivers and other kinds of medical service providers”, tells Janne Matikainen, Production and Logistics Manager at Lymphatouch.
In addition to negative pressure technology, LymphaTouch® has a mechanical high frequency vibration feature. It can be applied to several treatment areas, like lymphedema, preoperative and postoperative swelling and scarring, fascial tightness, muscle maintenance, active recovery, pain management and improving joint functionality.
“Our device is designed to support the lymphatic drainage. The negative pressure generated by the device helps to improve lymphatic circulation in the treated area which reduces pain and swelling as well as improves the functionality of the joints”, Matikainen explains and continues:
“The treatment areas are, for example, oncology, orthopedy and neurology. Additionally, our devices are used for muscle care and treating sports injuries of athlete’s.”
The first version of the device was launched already in 2009. The device the company is selling nowadays is the third version of it, and it has marketing authorizations both in Europe and in USA.
LymphaTouch® devices are manufactured at Innokas Kempele factory
The cooperation between Innokas Medical and Lymphatouch begun already in 2008 when Innokas co-created the first device version together with Lymphatouch and continued as their contract manufacturing partner. Promoting peoples’ well-being together by this cooperation has continued to present day as well; LymphaTouch® devices are manufactured at Innokas Kempele factory also nowadays.
“Innokas was chosen to our contract manufacturing partner because of the long-term, successful partnership we have had together. Innokas’ employees are professionals in the field and if there have been some changes and fixes that needed to be handled, Innokas’ performance has been efficient and the problems have been fixed effectively”, Matikainen tells.
”We see that Innokas has been able to deliver us the added value through their full-service offering, as well”, he continues.
By this, Matikainen means the different levels of the cooperation: in addition to manufacturing the devices for Lymphatouch, Innokas has took part in the design and development work of the latest device version by taking both manufacturability and component selection matters into account. Additionally, Innokas manufactured the prototypes before the actual design transfer. Matikainen says that they have been especially thankful for Innokas’ very strong quality and regulatory experience.
“As a global player, Innokas is a great co-operation company for our kind of smaller company also in the longer term”, Matikainen ponders.
”In addition to design and development as well as manufacturing services, Innokas Medical’s very strong quality and regulatory experience is something that has bring us added value. For example, the external audits related to our product have gone very well by now. Additionally, we’ve been able to have professional QA&RA related services from Innokas, which have helped us with new market acquisitions. We’ve been very satisfied with the cooperation with Innokas”, he concludes.
For more information, please contact:
Mikko Kangas, Business Development Director, Innokas Medical Ltd.: mikko.kangas@innokasmedical.fi
Janne Matikainen, Production and Logistics Manager, LymphaTouch: janne.matikainen@lymphatouch.com
For more information about the product, please go to:
https://www.lymphatouch.com/fi/