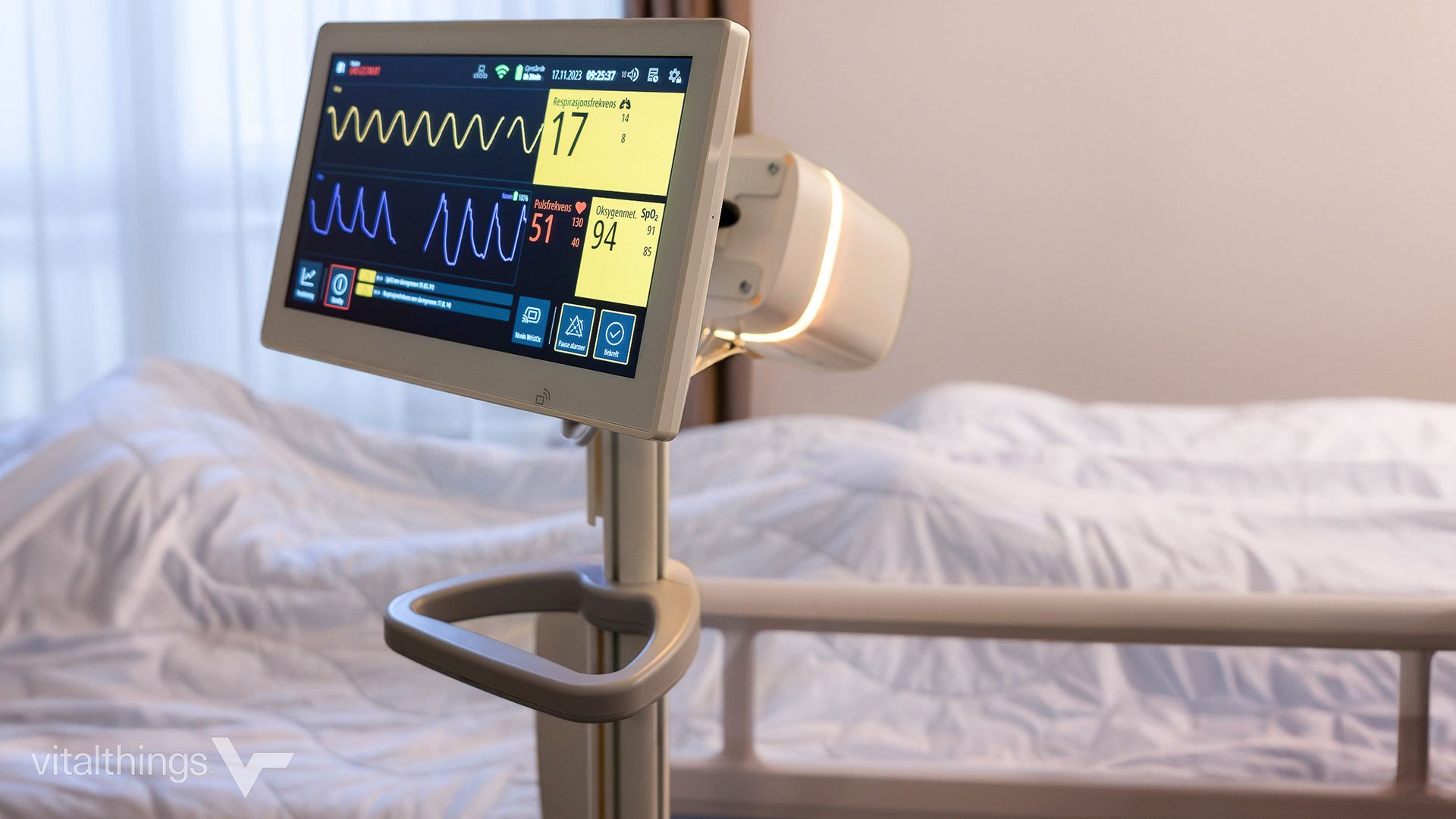
Complex product development projects are dynamic and require adaptability to maintain control over timelines and budgets while building the winning product. Teaming up with an experienced partner provides crucial expertise and adaptability during key phases, such as product specification and productization. Effective communication strategies ensure coordinated success throughout. This is how Innokas' collaboration with the award-winning HealthTech company Vitalthings led to the development of MDR-approved Class IIb medical device for contactless respiratory monitoring to be used in hospitals and healthcare institutions.
SUMMARY
- Innokas was chosen to partner with Vitalthings to develop the Guardian M10, a contactless respiratory monitoring device, and address regulatory compliance, technological complexity, and time-to-market pressures.
- Continuous communication and efficient transfer of responsibilities ensured steady progress, with steering group meetings and keeping a consistent core team.
- The Guardian M10 obtained MDR certification as a Class IIb device within nine months, with Innokas playing a pivotal role in product specification, productization, and transitioning to manufacturing.
Initiating an automated patient monitoring system development with partner guidance
Vitalthings is a Norwegian Healthtech company founded in 2014 specializing in proactive, contactless health monitoring for use in primary healthcare, hospitals, and research. Working to accomplish this, they have put their excellence into developing new and innovative patient monitoring concepts. Their product, Guardian M10, is set to ease the work of healthcare professionals by allowing continuous, and precise contactless monitoring of the patient's respiratory rate. Vitalthings is known, for example, from winning a competition set by St. Olav's hospital to determine their partner to create the solution of the future for measuring vital parameters, surpassing many other well-known players in the industry.
Partnership with Innokas
Innokas was brought in as a partner to relieve and address regulatory compliance, technological complexity, and time-to-market pressures. The partnership began in January 2021 with Vitalthings’ Guardian M10 project’s pre-study. Back then, the five-person project team consisted of a project manager, QARA Specialist, HW-, mech- and software engineers. The pre-study included drafting the initial design inputs and regulatory framework, gap analysis between current product and medical device, and concept design and medical device project planning in tight collaboration with Vitalthings internal project team and stakeholders.
Adapting to growth and expansion of the product development
Innokas guided the conception phase by asking relevant questions. In the project's early stages, numerous potential directions came under consideration. Some of the many questions presented were, for example, whether the product was going to be stand-alone ME equipment or multifunctional patient monitor. Each decision entails further questions. If the product was going to be a patient monitor, would it be for spot check or continuous monitoring, for example. Furthermore, it needed to be discussed if the product would be fixed, mobile or portable. Innokas offered insights into the implications of each decision for the product lifecycle and provided guidance on meeting regulatory demands. “We explored various ideas through studies to assess their suitability for the project,” says Teemu Uitto, project manager from Innokas who worked on the Guardian M10 project.
Project expansion and communication
The basic idea of the product concept was ready in early March 2021, but the concepting continued over the summer vacation period and during this time, the MD classifications and user needs inputs were defined, and different clinical solutions were studied, such as HL7 group of standards and integrating the device into hospital nursing monitors. The actual design project phase was kicked off at the end of August with Innokas’ strong participation. The project's direction had been determined; to develop a contactless respiratory monitor for use in hospitals and other healthcare institutions. Total Innokas team members in the project expanded from 5 to 9 people (adding usability engineer, ID design, SW developer and QARA consultants as well as clinical evaluation specialists from Innokas). Later a UX designer was added for concepting User interface as well as another HW developer because the number of PCBs increased. Innokas QA/RA experts were responsible for overseeing a wide range of the project's regulatory objectives, including supporting management of clinical investigation.
Efficient communication strategies
The team was expanded mainly to address the numerous changes in the project, along with the maturation of the requirements and user needs. However, there were generally no communication issues with people who joined the team later, and the transfer of responsibilities was kept efficient throughout. “We maintained continuous communication and conducted steering group meetings as often as necessary to keep everyone informed of the latest developments, with the customers CEO and project manager participating alongside our business developer,” Teemu says about the back and forth between Innokas and Vitalthings. “Additionally, maintaining a consistent core team throughout the project has facilitated much smoother progress.”
Successful collaboration and project completion
Experts from Vitalthings were happy with the quality of the collaboration. “We have had a good experience with Innokas and their regulatory and quality management services on the Guardian M10. Innokas also assisted us in moving our production to Tallinn, which has been an excellent process,” shares Sindre Holme, Head of Marketing and Communications at Vitalthings.
“For the Innokas choice of technologies, Jira and Confluence have worked very well on this project. At Vitalthings, we already use this software to document our processes. It has worked well in this case as well, not least when it comes to risk management and validation towards notified body. Overall, using Jira and Confluence has been a very smooth process,” Sindre continues about the technologies utilized in the project.
Mutual understanding and respect in partnership
“Our customer possesses strong competence in designing and manufacturing electromechanical devices,” Teemu affirms. “Thus, they had clear knowledge of what must, can, and cannot be done,” he continues. The clear and concise communication has benefitted the project, and the partnership has been characterized by mutual understanding and respect. Sindre agrees with this sentiment. “The project management on the Guardian M10 project has overall been a good experience. Especially the collaboration with Teemu and the NPI project manger Tebe Jürgenson, who are both very skilled people.”
The project has been successfully completed and the Guardian M10 has obtained MDR certification as a class IIb device in nine months. During the critical product specification and productization phase, Innokas assumed a pivotal role, particularly as Vitalthings lacked the requisite human resources for design and development. Responding swiftly to the demand, Innokas was able to expand the team with its own experts. Additionally, during the transition to the manufacturing phase, Innokas adeptly scaled down the team as required. Cooperation with Vitalthings continues on their new device project.
Innokas is a growing and agile family-owned technology company with 30 years of experience in highly demanding technology and have worked on an exceptionally wide range of customer projects, from Healthtech to other complex smart devices and solutions that help improve the quality of people’s lives. Our service portfolio includes manufacturing, engineering, software, and QA&RA, all complementing each other. In need of agile product development journey for safety- and quality-critical solutions, contact us for a chat.
More information about Vitalthings and their solution Guardian M10
Project Manager

Teemu Uitto
Head of Software